Pheromone-induced responses are mediated primarily by the vomeronasal organ (VNO). The VNO, also known as "Jacobson's organ", is part of an accessory olfactory system. It is present in a variety of non-human vertebrates but its existence in the human has been open to question until recently.
Historical Perspective
The VNO was first discovered by Ruysch (1703), a military doctor, in a soldier with a facial wound. The organ was named after Jacobson who published his findings on animals, but not humans, in 1811. Various other investigators have published studies; Potiquet (1891) observed the VNO in 25% of 200 adult humans, Pearlman (1934) described its occurrence in many animals, in human embryos and mentioned that it may be found occasionally in the adult human. (more detail can be found in Moran, Monti-Bloch, Stensaas and Berliner (1994)).
It was work published by Crosby and Humphrey (see below) that put the functional presence of the VNO in the human adult into doubt. In a standard anatomical textbook, "Correlative Anatomy of the Nervous System" by Crosby, Humphrey and Lauer (1962), we read the following; "In some human embryos, an accessory olfactory bulb, with complete representation of layering from the glomerular through the mitral cell layer, is to be found on the dorsomedial aspect of the main olfactory bulb (Humphrey, 1940). The accessory bulb receives the vomeronasal (or Jacobson's) nerve from the vestigial vomeronasal organ, which has also been demonstrated in the human embryo (McCotter, 1912). The vomeronasal nerve and its associated bulb vary in size and may be absent in the human embryo (Humphrey, 1940; Macchi, 1951). This variability appears to be characteristic of these structures in certain other adult primates (Lauer, 1945, 1949) Jacobson's organ and nerve and the accessory olfactory bulb are well developed in certain submammals and some subprimate mammals (Crosby and Humphrey, 1939). They are not present in adult man (Crosby and Humphrey, 1941). In human development these structures are merely ontogenetic signs of the persistence of phylogenetic structures and are without function".
Largely as a result of the above observations the existence of the VNO in humans has been disregarded. The consensus has been that, if it exists at all, it is vestigal and not functional. As recently as 1991 a paper by Meredith asserted that the accessory olfactory system is not present in humans or other old-world primates. This disregard for the existence of the VNO in adult humans is exemplified by the fact that the area containing the VNO is often removed during plastic surgery on the nose.
In 1980, Kreutzer and Jafek documented the early morphogenesis of the vomeronasal organ in the human embryo and early foetus. Later, in 1985, Johnson et al. examined 100 human adults specifically for the presence of the opening of the vomeronasal organ on the antero-interior part of the nasal septum. This was found in 39% of patients. Twenty seven human septums were removed post mortem, and vomeronasal structures were found in 70% of these. However, histological examination failed to reveal any evidence that these organs were functional (Johnson et al., 1985). In the same year, Nakashima et al. studied the VNO and nerves of Jacobson in a 28-week human foetus. They described the structure of the organ and traced the vomeronasal nervealong the nasal septum and through the cartilaginous cribriform plate with the olfactory nerve. While they found evidence for the existence of receptors in the vomeronasal epithelium similar to those in the olfactory epithelium, they suggested that the lack of intraepithelial blood vessels and mitotic figures in the epithelium indicated that the organ was undergoing degeneration at 28 weeks of gestation (Nakashima, Kimmelman and Snow, 1985). Since 1985 there has been a steady accumulation of evidence demonstrating the presence of the VNO in most, if not all, humans (Moran et al., 1985, 1991; Johnson et al., 1985; Stensaas et al., 1991; Garcia-Velasco and Mondragon, 1991, and reviewed in Moran et al., 1995).
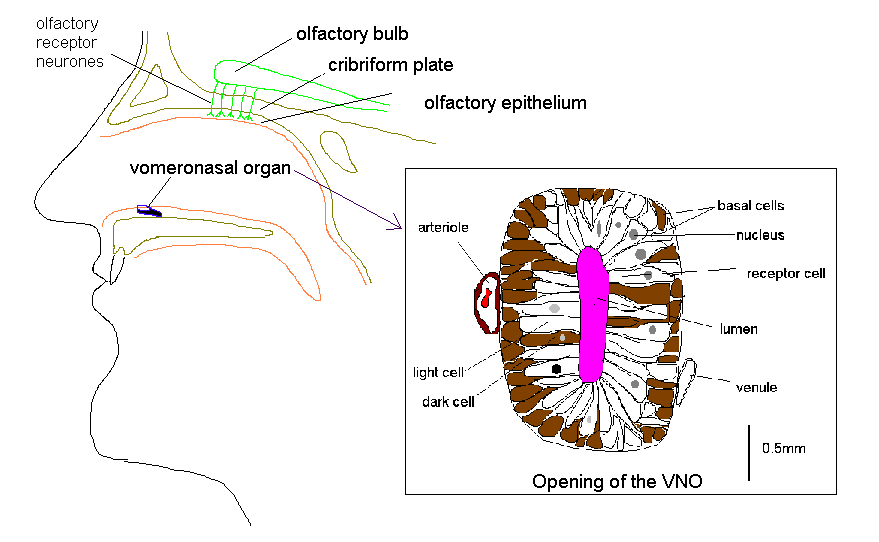
The VNO is located bilaterally on the anterior third of the floor of the nasal septum. It opens into the nasal cavity by a pit which varies in size from 0.2 to 2 mm situated 1-2 cm from the posterior margin of the nostril.
VNO Structure
The VNO is lined by a pseudostratified, columnar epithelium some 60um in depth that lies on a thick basement membrane. The vomeronasal neuroepithelium contains three morphologically distinct cell types; basal cells, dark cells and light cells. The basal cells are small, polygonal, dark-staining cells, measuring 6um in diameter. The dark cells are tall, slender, columnar cells with densely staining cytoplasm. The light cells are tall, columnar cells that, like the dark cells, extend from the basement membrane to the free surface of the epithelium. They differ from the surrounding cells of the respiratory epithelium in the nose by having no cilia (Moran et al, 1995).
VNO Receptors
The vomeronasal organ in the human foetus from 12 to 23 weeks is lined by a pseudostratified epithelium, with neurone-specific enolase (NSE) positive cells which resemble olfactory receptors. However, at 36 weeks the organ was lined by a respiratory epithelium and did not show any receptor-like cells, although there were some pear-shaped NSE-positive cells in the upper part of the respiratory epithelium of unknown significance (Boehm and Gasser, 1993). It was suggested that, during the early foetal period, the VNO could have some, as yet unknown, sensory function (op. cit.).
In contrast, calbindin, a 28Kd protein that has been immunolocalised to VNO receptor cells, was found to be expressed in humans, firstly in a male neonate and secondly an adult female suggesting the presence of receptors during early development and in the adult (Johnson, Eller and Jafek, 1994)
Whether or not the VNO continues to express receptor cells or neuronal markers in the adult, the structure itself continues to expand according to Smith et al. (1997). When comparing the increase in length of the VNO in the developing foetus to the length in the adult human, they concluded that further prenatal or postnatal size increase must occur.
VNO - Functionality
Compounds occurring naturally on the human skin were found to cause a local depolarisation when applied directly to the VNO (Monti-Bloch and Grosser, 1991). The nature of these compounds was not disclosed. This depolarisation had the characteristics of a receptor potential. Furthermore these compounds did not cause a response from the olfactory epithelium and, olfactory stimulants (e.g. cineole) had no effect on the VNO. Using the same compounds sexual dimorphism was demonstrated in their effect on electrodermal activity (Monti-Bloch et al, 1994). These compounds were subsequently revealed to be 16-androstenes and estrenes (Berliner, 1993; 1994). The androstenes have been previously isolated from human sweat (secreted by the axillary apocrine glands) (Gower et al., 1985).
Another "vomeropherin", pregna-4,20-diene-3,6-dione (PDD), caused evoked potentials in the VNO and also changed gonadotropin pulsatility in males, resulting in a reduced level of luteinizing hormone (Berliner et al, 1996) and testosterone (Monti-Bloch et al, 1998). In addition, PDD decreased respiratory frequency, increased cardiac frequency and caused event-related changes of electrodermal activity in EEG pattern (Berliner et al, 1996).
Is The Adult Human VNO Vestigial?
The overwhelming anatomical evidence suggests that the in the adult human the VNO is vestigial (Meredith, 2001). There is no evidence for any anatomical connection from the VNO to the brain. Furthermore, the channel responsible for pheromone transduction in lower mammals, TRP2 (a non-selective cation channel), is a pseudogene in humans. However, this does not mean that human pheromones do not exist. mRNA for a vomeronasal receptor, hV1RL1, has been found in the human olfactory epithelium (Rodriguez & Mombaerts, 2002).
Human Pheromones
When pheromones were first discovered in the late 1950s they were defined as “..substances which are secreted to the outside by an individual and received by a second individual of the same species, in which they release a specific reaction, for example, a definite behaviour or a developmental process” (Karlson and Luscher, 1959).
Following the pioneering work on menstrual synchrony by McClintock (1971), Stern and McClintock (1998) have shown that odourless axillary compounds from the armpits of women in the late follicular phase of their menstrual cycles accelerated the preovulatory surge of luteinizing hormone of recipient women and shortened the menstrual cycles. Axillary compounds from the same donors which were collected later in the menstrual cycle (at ovulation) had the opposite effect: they delayed the luteinizing hormone surge of the recipients and lenghtened their menstrual cycle (Stern and McClintock, 1998).
The work by McClintock is very supportive of the existence of a chemical signal in axillary secretion - a pheromone - but it does not prove the existence of a functional VNO.
Androstadienone
Androstadienone is the current best candidate we have for a human pheromone. Is is a component of human (in particular male) secretions. Does androstadienone meet Karlson & Luscher (1959) "pheromone" criteria? In human studies in which androstadienone had access to the olfactory mucosa both physiological and psychological effects have been reported (Jacob and McClintock, 2000; Jacob, Hayreh and McClintock, 2001; Jacob, Garcia, Hayreh and McClintock, 2002 Bensafi et al., 2003; Lundstrom et al., 2003; Bensafi et al 2004a, 2004b; Cornwell et al., 2004). While none of these changes can be regarded as the behavioural changes required for a compound to qualify as a pheromone, Savic et al. (2001) demonstrated that androstadienone activated the hypothalamus in a gender-specific manner (it activated the hypothalamus in women but not men). Compared with other odorous substances, androstadienone activated the anterior part of the inferior lateral prefrontal cortex (PFC) and the superior temporal cortex (STP) in addition to olfactory areas (Gulyas et al., 2004). The PFC and STP have been implicated in aspects of attention, visual perception and recognition and social cognition.
The definition of a pheromone (see above, Karlson and Luscher 1959) was later modified to include a mutual benefit requirement (Rutowski, 1981). In this context it is possible to see such mutual benefit if the pheromone leads, for example, to choice of partner on the basis of their HLA-dependent odortype to increase immune diversity (Ober et al., 1997, 1999) and lower miscarriage rates (Ober et al., 1998) or if the pheromone participates in the mother-infant bonding process (Schleidt, 1992). On its own it is unlikely to be able to achieve these outcomes but, as recently reported by Coureaud et al (2004), a pheromone can act as a reinforcing agent or a one-trial conditioning agent in which the presence of the pheromone converts a second odour (that of the partner or infant) into a conditioned stimulus.
Odor and Major Histocompatibility Complex
Mice can distinguish one another by odour. This odour is genetically determined and partly specified by the H-2 major histocompatibility complex (MHC) (Yamazaki et al, 1979; 1994) on chromosome 17 (Schellinck et al, 1993). There are however other genetic loci coding for odortypes found on the nonrecombining part of the Y chromosome (Schellinck et al, 1993). These odors are excreted in the urine and may play a part in pregnancy block (the Bruce effect), aggression and other mouse social behaviours.
The human equivalent of the MHC locus is the human leucocyte antigen (HLA). Numerous studies have been carried out with humans that suggest that axillary odour contains enough chemical differences in its odour profile to allow for discrimination between individuals. It has been hypothesised that at least some of this individual specific odour may be under the control of human leucocyte antigen (HLA) genes (Preti, Spielman and Wysocki, 1997). Studies have shown that women prefer those male odours that have HLA types different from their own - a preference that was reversed when those women doing the rating were taking oral contraceptives.
Supporting evidence for the existence of individual-specific odour comes from studies in which mothers have identified their own newborn infants from the smell of a previously worn T-shirt (Schaal et al, 1980). In turn, infants prefer breast or axillary pads from their own mothers, distinguishing the odour from other kin (Schaal et al, 1991).
Summary
Pheromones are well established in many species but the their existence in humans is still controversial. Much of the problem lies with the fact that the original definition of a pheromone cannot allow for volitional control over the biological response. Humans have a will of their own and can refuse the biological imperative. How then do you determine if a chemical signal is altering human behaviour? The experiments that have attempted to do this, e.g. by measuring autonomic responses, have had extremely variable results. The results in one lab are different to those in another. This implies that the are too many uncontrolled variables. Humans are obstinately different. They respond differently depending upon whether the experimenter is male or female for instance!
While the behavioral changes induced by axillary secretions are supportive evidence, they have not convinced everyone in the field - measuring menstrual synchrony is a process fraught with problems. Receptors (or at least their mRNA) for pheromones have been found in the human olfactory epithelium, but we are still waiting for someone to demonstrate the presence of a pheromone receptor protein expressed in the surface membrane of an olfactory receptor neuron and that responds to a ligand (a potential pheromone) - as has been demonstrated for the olfactory receptors.










































